Haber-Bosch process
Industrial synthesis of ammonia (NH₃) from atmospheric nitrogen and hydrogen under high pressure with an iron catalyst. Without it, only about 4 billion humans could be fed.
Molecular synthesis through controlled chemical reactions
Key reaction
Operating conditions
- Temperature
- 400-450°C
- Pressure
- 150-300bar
- Catalyst
- Fe / Fe₃O₄ promu K₂O + Al₂O₃
- Phase
- gas
How it works
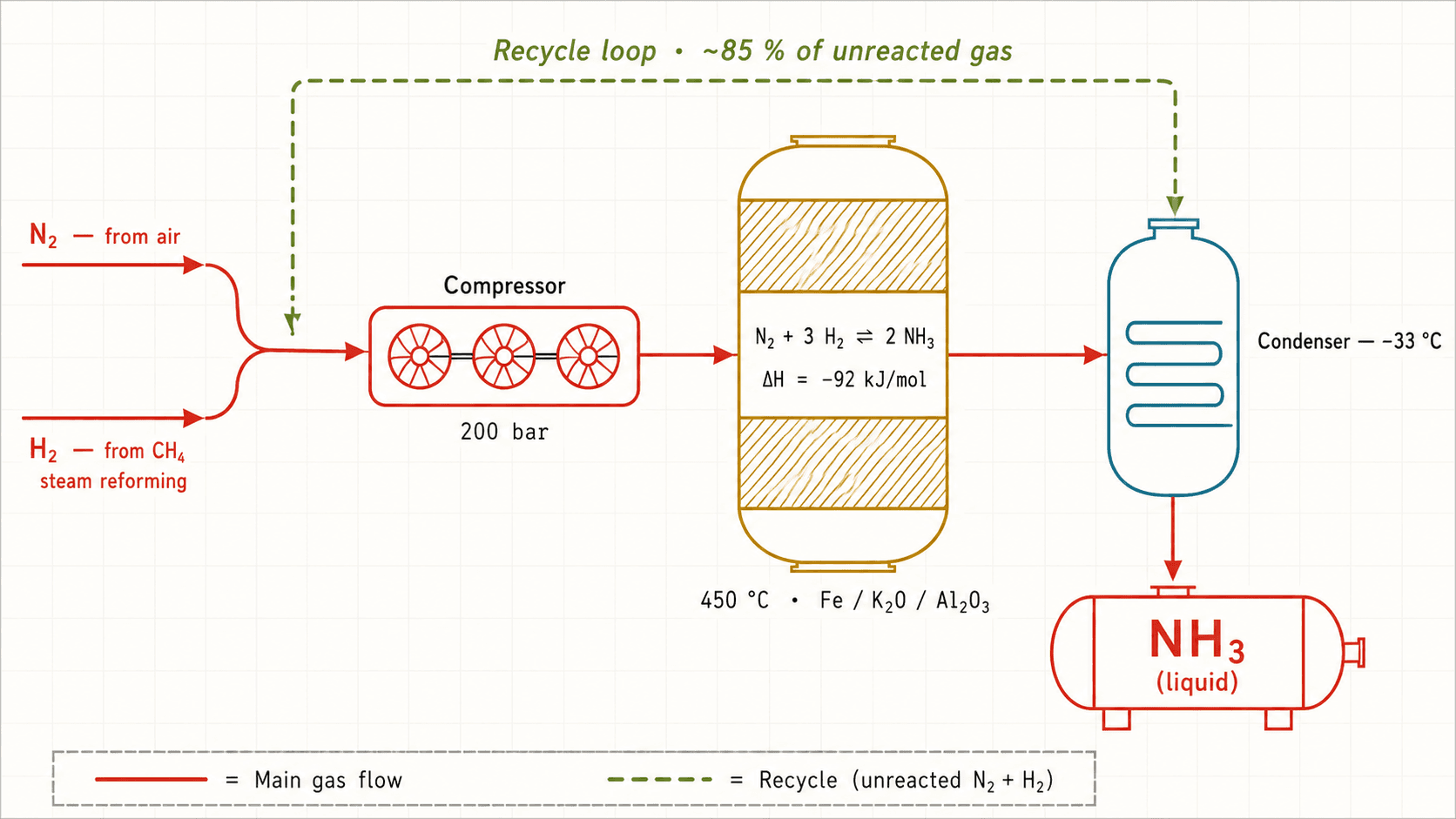
How it works
Key components
The role of each main part, and the elements / compounds it involves.
Multi-stage compressor
Compresses the N₂ + H₂ mixture up to 150-300 bar to shift the equilibrium toward NH₃ formation.
Compression is staged (typically 4 to 6 stages) with inter-stage cooling to keep temperature rises in check. Each stage raises pressure by a factor of ~2-3. It is the most energy-intensive part of the process — about 10-15 % of an ammonia plant's total energy budget.
150-300 bar · 4-6 étages · refroidissement inter-étage
See also :n2h2Iron-catalyst reactor
Heart of the process. The gas mixture flows through the catalyst bed where ~15 % of the N₂ and H₂ combine into NH₃.
The catalyst is finely divided metallic iron (Fe / Fe₃O₄), promoted by K₂O (which activates N₂ dissociation) and stabilized by Al₂O₃ (prevents sintering at high temperature). The reactor runs at 400-450 °C — a compromise: hotter speeds up kinetics but shifts the equilibrium back toward reactants (exothermic reaction). Per-pass conversion is capped at ~15 %, hence the recycle loop.
400-450 °C · 200 bar · catalyseur Fe / K₂O / Al₂O₃ · ~15 % conv./passage
See also :n2h2nh3fekalRefrigerated condenser
Liquefies the ammonia formed at the reactor outlet, separating it from unreacted N₂ and H₂.
At 200 bar, NH₃ condenses around −20 to −30 °C while N₂ and H₂ remain gaseous (boiling points −196 and −253 °C). Liquid NH₃ is drawn off at the bottom and stored; non-condensable gases are sent back to the compressor through the recycle loop. This phase-change separation is what makes the whole process economical.
−20 à −30 °C · 200 bar · NH₃ liquide / N₂-H₂ gazeux
See also :nh3High-pressure recycle loop
Sends the ~85 % of unconverted gas back to the reactor to reach overall conversion > 97 %.
Without recycling, losing 85 % of the feed on each pass would kill the economics. The loop keeps the mixture under pressure — avoiding decompression and recompression saves a huge amount of energy. A small purge bleeds out inert gases (Ar, residual CH₄) that would otherwise accumulate.
≈ 85 % du débit gazeux · purge inertes ~2 %
Physical and chemical principles
The fundamental laws that make this process possible — and the constraints they impose.
Le Chatelier's principle (pressure-shifted equilibrium)
The reaction goes from 4 moles of gas (1 N₂ + 3 H₂) to 2 moles (2 NH₃) — the product side has fewer moles. Raising pressure forces the system to minimize volume, hence to form more NH₃. That's exactly why the process operates at 200 bar: without it, the equilibrium is too unfavorable even at high temperature.
N₂ + 3 H₂ ⇌ 2 NH₃ (Δn = −2 mol gaz)Heterogeneous catalysis (breaking the N≡N triple bond)
The N≡N triple bond (945 kJ/mol) is one of the strongest in chemistry. Without a catalyst, dissociating N₂ would require temperatures above 1000 °C. Iron chemisorbs N₂, weakens the N≡N bond, and lets H₂ (itself dissociated on the surface) add stepwise. K₂O boosts activity by donating electrons to iron; Al₂O₃ structures the support to prevent sintering.
N₂(ads) → 2 N(ads) → … → 2 NH₃Applies to components :reacteur-catalytiqueKinetic vs thermodynamic trade-off
The synthesis is exothermic (ΔH = −92 kJ/mol). At low temperature the equilibrium favors NH₃ but the reaction is too slow. At high temperature kinetics are fast but the equilibrium shifts toward reactants. 400-450 °C is the window where per-pass conversion stays decent (~15 %) while sustaining industrial throughput. The whole plant design revolves around that trade-off.
ΔH = −92 kJ/mol · K_eq décroît avec TApplies to components :reacteur-catalytique
Compounds involved
Output
World production
Main applications
- Nitrogen fertilizers (urea, nitrates)80 %
- Explosives and industrial nitrates8 %
- Plastics (nylon, polyurethanes)5 %
- Hydrogen carrier (emerging, maritime shipping)4 %
- Industrial refrigeration3 %
Decarbonization and current challenges
- Ammoniac vert : H₂ produit par électrolyse alimentée 100 % renouvelable (Yara Norvège, NEOM Arabie Saoudite)
- Capture-utilisation du CO₂ co-produit (CCUS, urée bas-carbone)
- Mini-usines décentralisées (~1-10 tNH₃/jour) pour fermes et petites communautés
- Catalyseurs ruthénium-baryum permettant 100 °C et 50 bar — encore en R&D
- Vecteur énergétique : NH₃ comme carburant maritime (IMO 2050) et stockage long terme d'H₂
Similar or competing processes
Related industrial processes — alternative chemistry, alternative technology.
- electrosynthese-nh3
Direct electrochemical synthesis of NH₃ from N₂ + H₂O — eliminates the Haber-Bosch step and H₂ production altogether. Still at lab scale, low yields.
- frank-caro
Historical process (1898) that produced calcium cyanamide CaCN₂ as a fixed-nitrogen carrier. Phased out in the 1920s as Haber-Bosch's far better energy efficiency took over.